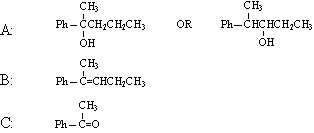
Answer Key for Exam I (unit I)
1) description and identification of peaks:
|
wavenumber (cm-1) |
intensity |
functional group (s) |
|
~3000 cm-1 ~1700 cm-1 |
Strong, broad Strong, sharp |
OH C=O |
2) draw the structures for A-C:
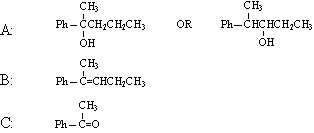
3) functional group(s):
a) alcohol
b) alkene
4) 1H NMR to differentiate between ketone, aldehyde and acid:
aldehyde: peak between 9-10 ppm
acid: peak between 10-12 ppm
ketone: no peak past 9 ppm
5) number of different types of protons:
|
a) 5 |
b) 3 |
c) 1 |
d) 3 |
|
e) 3 |
f) 1 |
g) 1 |
h) 5 |
6) a structure of C6H3Br3:
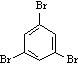
7) the correct structure: b
reasons for incorrect structures:
not (a) b/c there is no triplet or quartet in the 1H spectrum
not (c) b/c there is only OCH types in the structure given
not (d) b/c there is no aldehyde peak around 9-10ppm in the spectrum; also there is 4 different types of protons in this structure.
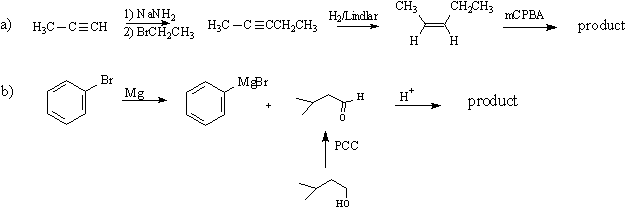
8) draw the structures or conditions for A-E:
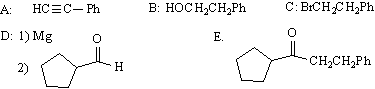
9) proposed structures (give two out of three answers):
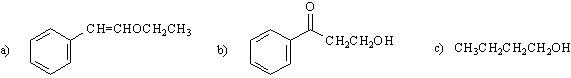
10) Note: there are many answers possible for the synthesis problems.
1
H NMR of compound (a)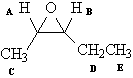
|
diff. protons |
Type of protons |
chemical shift (ppm) |
number of protons (integration) |
splitting pattern (m.q,t,d,s) |
|
A |
OCH |
3.5-4.5 |
1 |
~quintet |
|
B |
OCH |
3.5-4.5 |
1 |
~quartet |
|
C |
CH |
.8-1.5 |
3 |
Doublet |
|
D |
CH |
.8-1.5 |
2 |
Quintet |
|
E |
CH |
.8-1.5 |
3 |
triplet |
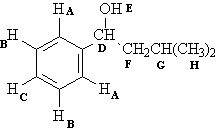 1H NMR of compound (b):
1H NMR of compound (b):
|
diff. protons |
Type of protons |
chemical shift (ppm) |
number of protons (integration) |
splitting pattern (m.q,t,d,s) |
|
A |
Ar-H |
7-8 |
2 |
Doublet |
|
B |
Ar-H |
7-8 |
2 |
Triplet |
|
C |
Ar-H |
7-8 |
1 |
Triplet |
|
D |
OCH |
3.5-4.5 |
1 |
Triplet |
|
E |
OH |
.5-5 |
1 |
Singlet |
|
F |
CH |
.8-1.5 |
2 |
Triplet |
|
G |
CH |
.8-1.5 |
1 |
Nonet |
|
H |
CH |
.8-1.5 |
6 |
doublet |